Research
A Novel Biomechatronic Interface Based on Wearable Dynamic Imaging Sensors

The problem of controlling biomechatronic systems, such as multiarticulating prosthetic hands, involves unique challenges in the science and engineering of Cyber Physical Systems (CPS), requiring integration between computational systems for recognizing human functional activity and intent and controlling prosthetic devices to interact with the physical world. Research on this problem has been limited by the difficulties in noninvasively acquiring robust biosignals that allow intuitive and reliable control of multiple degrees of freedom (DoF). The objective of this research is to investigate a new sensing paradigm based on ultrasonic imaging of dynamic muscle activity.We have shown the feasibility of using inexpensive ultrasound imaging probes to spatially resolve and visualize dynamic activity of different functional compartments in the forearm muscles through image processing in able-bodied subjects as well as transradial amputees.
In an ongoing study, we are collaborating with the MedStar National Rehabilitation Hospital and the Washington DC Veterans Affairs Medical Center to recruit amputee subjects. Subjects utilize our prototype sonomyographic control systems to control a virtual prosthetic hand and interact with a virtual reality environment. They are being trained to control various degrees of freedom individually and in combination using a biofeedback paradigm. We will then evaluate the performance for completing different tasks related to activities of daily living, with and without increased cognitive load.
The proposed research methods are broadly applicable to assistive technologies where physical systems, computational frameworks and low-power embedded computing serve to augment human activities or to replace lost functionality.
Funded by:
National Science Foundation
Closed-loop Hybrid Exoskeleton utilizing Wearable Ultrasound Imaging Sensors for Measuring Fatigue
The goal of this project is to develop an automated assistive device capable of restoring walking and standing functions in persons with motor impairments. Although research on assistive devices, such as active and passive orthoses and exoskeletons, has been ongoing for several decades, the improvements in mobility have been modest due to a number of limitations. One major challenge has been the limited ability to sense and interpret the state of the human, including volitional motor intent and fatigue. In collaboration with the University of Pittsburgh, we are designing a hybrid exoskeleton consisting of powered electric motors, as well functional electric stimulation to generate power from the subject’s muscles. We are using wearable ultrasound sensors to identify muscle fatigue and allocate control. The proposed project has the long-term potential to significantly improve walking and quality of life of individuals with spinal cord injuries and stroke. This framework can be widely applicable in a number of medical CPS problems that involve a human in the loop, including upper and lower extremity prostheses and exoskeletons, rehabilitation and surgical robots. The new control allocation algorithms relying on sensor measurements could have broader applicability in fault-tolerant and redundant actuator systems, and reliable fault-tolerant control of unmanned aerial vehicles.
Funded by:
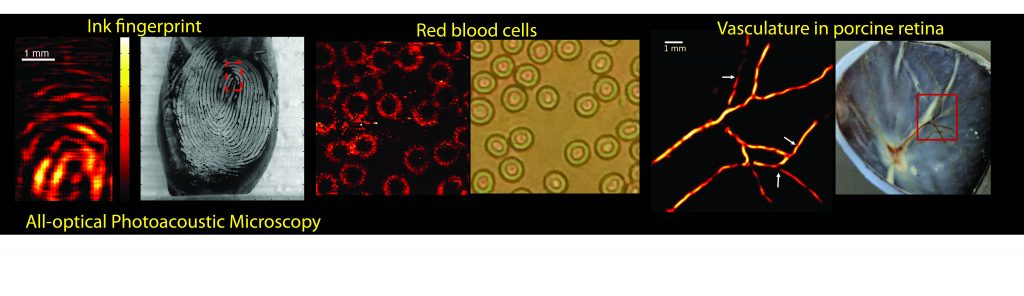
All-optical Photoacoustic Microscopy for Imaging the Retina
The aim of this project is to assess the feasibility of a novel, all-optical technique for frequency-domain photoacoustic imaging that employs a photorefractive crystal and potentially can provide anatomical and functional images of the retinal structures with fine-resolution. Detailed visualization of retinal substructures and the optic nerve is critical for evaluation of ophthalmic diseases such as diabetic retinopathy, age-related macular degeneration and glaucoma. However, vascular function and local oxygen delivery also are relevant indicators of the diseased state.
We have developed a frequency-domain photoacoustic system that can potentially facilitate video-rate functional microscopy. We are working to demosnmtrate the ability of our techniques to depict retinal/choroidal micro-circulation and the retinal-pigmented epithelium microstructure as well as functional changes related to altered oxygen saturation. Images obtained using the proposed methods will be compared with optical fundus images and histology- based ground truth. Successful completion of the proposed study will establish a basis for developing a versatile system for ophthalmic imaging that can be employed in pre-clinical research using a variety of animal models, and ultimately in clinical imaging.
Funded by:
National Institutes of Health
Asymptomatic Carotid Atherosclerosis and Correlates of Cognitive Function
Carotid artery plaques are known to cause stroke. Cognitive impairment is an insidious but poorly understood problem in patients with carotid plaques. Cognitive function describes how people perform mental processes such as thinking, learning and problem solving. Asymptomatic carotid plaques affect a significant portion of the population, including 1 million Veterans, who may be at risk for cognitive impairment. In this collaborative study with the VA Medical Center in Baltimore, MD, we are seeking to uncover the extent of cognitive impairment in Veterans with carotid stenosis who are currently labeled “asymptomatic”. Programs to prevent or mitigate cognitive impairment will depend on identifying the mechanisms by which this occurs. We are using sophisticated 3D ultrasound imaging techniques developed by our group to measure the structure and composition of plaques, number of particles breaking off from them, blood levels of chemicals that could disrupt them, and blood flow restriction to the brain from them. This will help identify patients at risk for cognitive impairment who may benefit from preventative measures and improve selection of patients to decrease unnecessary surgical procedures.
Funded by:
Department of Veterans Affairs
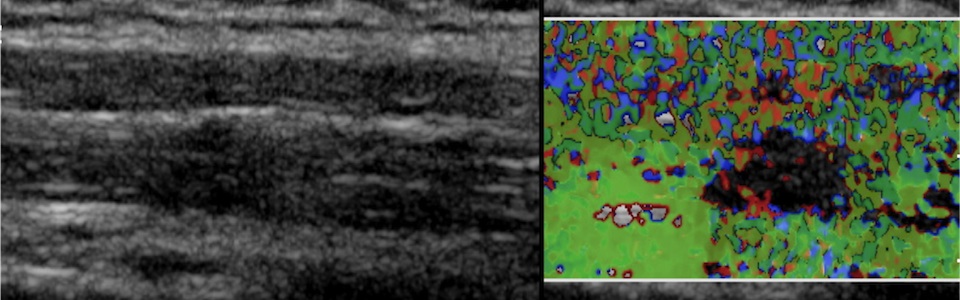
Understanding the Pathophysiology of Chronic Soft Tissue Pain Syndromes
Chronic pain affects approximately 100 million adults in the US and is associated with an annual cost of $560-635 billion. Our long-term goal is to identify which patients are at a higher risk of developing a complex difficult to treat chronic pain state, and tailor therapeutic interventions that are best suited for each patient’s unique clinical presentation. In previous work, we sought to understand the physical and physiological milieu of soft tissue (e.g., muscle, fascia, blood vessels) associated with active (i.e. spontaneously painful) myofascial trigger points (MTrPs) in patients with chronic neck pain, and to correlate these findings with clinical assessments. We showed that: 1) The mechanical tissue properties, vascular physiology and biochemical milieu of the affected soft tissue neighborhood of active MTrPs in patients with chronic neck pain are different compared to asymptomatic control subjects with/without palpable MTrPs; and 2) A physical perturbation caused by dry needle therapy, has a significant and measurable effect on the soft tissue environment of active MTrPs in symptomatic subjects. We are now continuing this work to understand the underlying mechanisms that lead to these soft tissue differences.
Funded by:
National Institutes of Health
Dynamic Imaging of Muscle Function using Ultrasound
Our lab has been investigating the use of ultrasound imaging as a method for studying the dynamic activity of skeletal muscle. This method is complementary to conventional electromyography using surface electrodes, and in some applications can provide significant benefits by overcoming the well-known limitations of surface electromyography in terms of limited specificity and poor signal to noise ratio. Real-time ultrasound imaging is uniquely suited for dynamic muscle function studies because it is cost-effective, portable and can be integrated with other measurements. However, lack of quantitative dynamic measures and challenges due to anisotropy of muscle have been barriers to widespread use of ultrasound. The proposed research is designed to overcome these barriers. The approach is to develop novel dynamic ultrasound imaging modes for quantifying anisotropic muscle kinematics, viscoelastic tissue properties and blood flow, and integrate these novel measures with conventional measures of tissue oxygenation, electrical activation, strength, and range of motion to characterize the underlying physiological systems.

Funded by:
National Science Foundation
In addition to its diagnostic capabilities, ultrasound can generate a variety of physical effects in tissue related to heating, radiation force (in situ “push”), and cavitation, which can be leveraged to produce a range of desired bioeffects. Ultrasound is particularly attractive because it is economical, portable, and nonionizing.
Ultrasound-based actuation and control of implantable bio-machines
The objective of this project is to develop ultrasound-based strategies for controlling thermally sensitive implantable drug-delivery devices. We have demonstrated that ultrasound can noninvasively heat biocompatible hydrogel components, which then rapidly and reversibly change their dimensions. This strategy can be used to incorporate and control mechanically moving structures such as valves, pumps, gears, etc., in a variety implantable devices. We are actively working on elucidating the underlying physics involved in this paradigm of localized drug delivery and translate the approach to address specific clinical needs such as localized chemotherapy of solid tumors. (Ordeig, O., S.Y. Chin, S. Kim, P.V. Chitnis, and S.K. Sia, An implantable compound-releasing capsule triggered on demand by ultrasound. Scientific Reports, 2016. 6: p. 22803)
Funded by:
National Science Foundation
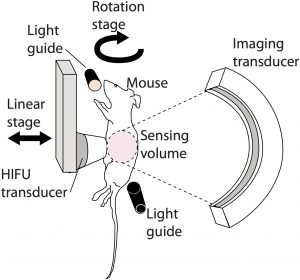
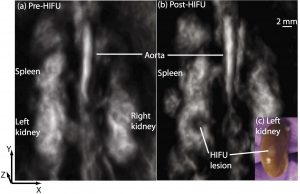
Photoacoustic monitoring and guidance of high-intensity focused ultrasound
High-intensity focused ultrasound (HIFU) can noninvasively heat diseased tissue to deliver localized hyperthermia. Such approaches offer viable alternative to invasive surgical procedures for treating tumors. However, noninvasive therapeutic modalities necessitate noninvasive and real-time monitoring of local temperature and tissue properties to ensure successful outcome while minimizing collateral thermal damage to surrounding healthy tissue. Photoacoustic imaging is one of the imaging modalities being researched for meeting this need. Photoacoustic-signal amplitude is linearly proportional to temperature. It also depends on local optical-absorption properties, which change when tissue undergoes thermal coagulation. (Chitnis PV, Brecht H, Su R, Oraevsky AA; Feasibility of optoacoustic visualization of high-intensity focused ultrasound-induced thermal lesions in live tissue. J. Biomed. Opt. 0001;15(2):021313-021313-5.)